Nuclear Physics and RadioactivityHard
Question
An electron collides with a fixed hydrogen atom its ground in its ground state. Hydrogen atom gets existed and the colliding electron loses all its kinetic energy. Consequence the hydrogen atom may emit a photon corresponding to the largest wavelength of the Calmer series. The min. K.E of coiling electron will be -
Options
A.10.2eV
B.1.9 eV
C.12.1 eV
D.13.6 eV
Solution
Excitation up to n = 3 is required so that visible length is emitted upon de - excitation
So required energy = 13.6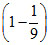 = 13.1eV
= 13.1eV
So required energy = 13.6
Create a free account to view solution
View Solution FreeMore Nuclear Physics and Radioactivity Questions
The compound unstable nucleus often decays in accordance with the following reaction + other particles. In the nuclear r...A radioactive nucleus decay as follows :- X X1 X2 X3 X4, if the atomic number and the mass number of X are 72 and 180 th...An electron is in an excited stable in hydrogen - like atom. It has a total energy of -3.4 eV. If the kinetic energy of ...The half life of a radioactive isotope is 2 hour, after which times it remains 1/64 times of its initial value...A $^{22}_{10}$Ne nucleus, after absorbing energy, decays into two $\alpha$-particles and an unknown nucleus. The unknown...