CircleHard
Question
Which of these is not a monomer for a high molecular mass silicone polymer ?
Options
A.PbSiCl3
B.MeSiCl3
C.Me2SiCl2
D.Me3SiCl
Solution
Silicons are organo-silicon polymer containing 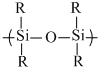 linkage. Since Me3SiCl contain only one Cl therefore it can′t form high molecular mass silicon polymer
linkage. Since Me3SiCl contain only one Cl therefore it can′t form high molecular mass silicon polymer
It act a chain terminating organo silane
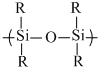 linkage. Since Me3SiCl contain only one Cl therefore it can′t form high molecular mass silicon polymer
linkage. Since Me3SiCl contain only one Cl therefore it can′t form high molecular mass silicon polymerIt act a chain terminating organo silane
Create a free account to view solution
View Solution FreeMore Circle Questions
The gradient of the tangent line at the point (a cos α, a sin α) to the circle x2 + y2 = a2, is-...The locus of centre of the circle which cuts the circle x2 + y2 = k2 orthogonally and passes through the point (p,q) is ...The parametric coordinates of any point on the circle x2 + y2 - 4x - 4y = 0 are -...A pair of tangents are drawn from the origin to the circle x2 + y2 + 20(x + y) + 20 = 0. The equation of the pair of tan...Circle x2 + y2 − 4x − 8y − 5 = 0 will intersect the line 3x − 4y = m in two distinct points, if ...