Nuclear Physics and RadioactivityHard
Question
Suppose the potential energy between electron and proton at a distance r is given by - 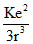 Application of Bohr′s therapy to hydrogen atom in this case shown that
Application of Bohr′s therapy to hydrogen atom in this case shown that
Options
A.energy in the nth orbit is proportional to n6
B.energy is proportional to m-3(m = mass of electron)
C.energy in the nth orbit is proportional to n-2
D.energy is proportional to m3 (m = mass of electron)
Solution
U = - 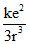 ; F =
; F = 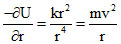
mv2 r3 = ke2; m2v2r2 =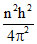
r =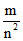 (constant), Also V2 ∝
(constant), Also V2 ∝ 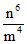
U =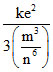 , k.e. =
, k.e. = 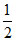 mv2 ∝
mv2 ∝ 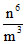
U ∝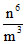 , T.E. = U + K.E. ∝
, T.E. = U + K.E. ∝ 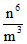
then total energy ∝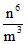
mv2 r3 = ke2; m2v2r2 =
r =
U =
U ∝
then total energy ∝
Create a free account to view solution
View Solution FreeMore Nuclear Physics and Radioactivity Questions
At some instant two radioactive substance are having amount in ratio of 2 : 1. Their half lives are 12 hrs and 16 hrs th...Who discovered the nuclear fission :-...The radioactivity of a sample is R1 at a time T1 and R2 at a time T2. If the mean life of the specimen is T, the number ...Two electrons are moving with the same speed v. One electron enters a region of uniform electric field while the other e...7/8 fraction of a sample disintegrates in t time. How much time it will take to disintegrete 15/16 fraction :-...