Nuclear Physics and RadioactivityHard
Question
The electron in a hydrogen atom makes a transition from n = n1 to n = n2 state. The time period of the electron instate n1 is eight times in state n2. The possible values of n1 and n2 are
Options
A.n1 = 8, n2 = 1
B.n1 = 4, n2 = 1
C.n1 = 4, n2 = 2
D.n1 = 2, n2 = 4
Solution
Time period T = 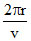 ∝ n3 But T1 = 8T2 ⇒ n13 = 8n23 ⇒ n1 = 2n2.
∝ n3 But T1 = 8T2 ⇒ n13 = 8n23 ⇒ n1 = 2n2.
Create a free account to view solution
View Solution FreeMore Nuclear Physics and Radioactivity Questions
Mass number and atomic number of radioactive material are respectively 232 and 90. After disintegration mass number is 2...The probability of nucleus to decay in two mean lives is...If half life period of radium is 1600 years then its average life is -...The position of and are shown on the binding energy curve shown in figure.The energy released in the fusion reaction....An archaeologist analyses the wood in a prehistoric structure and finds that C14 (Half life = 5700 years) to C12 is only...