Nuclear Physics and RadioactivityHard
Question
A photon of energy 12.09 eV is completely absorbed by a hydrogen atom initially in the ground state. The quantum number of the excited state is
Options
A.4
B.5
C.3
D.2
Solution
Energy difference in hydrogen atom = 13.6 - 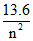 = 12.09 ⇒ n2 ≈ 9 ⇒ n = 3
= 12.09 ⇒ n2 ≈ 9 ⇒ n = 3
Create a free account to view solution
View Solution FreeMore Nuclear Physics and Radioactivity Questions
A boy playing on the roof of a 10 m high building throws a ball with a speed of 10 m/s at an angle of 30° with the horiz...Plutonium - decays with a half life of 24000 years. If plutonium is stored for 72000 years, then the fraction of plutoni...If elements principle quantum number n > 4 were no allowed in nature, the number of possible elements would be :...Ratio of initial active nuclei in two different samples is 2 : 3. Their half lives are one hour and two hours respective...Find the half-life period of a radioactive material, if its activity drops 1/32 th of its initial value in 20 years :...