Ionic EquilibriumHard
Question
When 0.002 mol of acid is added to 250 mL of a buffer solution, pH decreas by 0.02 units. The buffer capacity of the system is
Options
A.0.1
B.0.2
C.0.3
D.0.4
Solution
Buffer capacity = 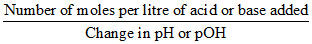
[Acid] =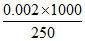 = 0.008 M
= 0.008 M
Buffer capacity =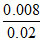 = 0.4
= 0.4
[Acid] =
Buffer capacity =
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
The curve in the figure shows the variation of pH during the course of titration of a weak acid, HA with a strong base (...0.1mol HCl is dissolved in distilled water of volume V then at limV→∞ (pH)solution is equal to...The best indicator for the detection of end point in titration of a weak acid and a strong base is :...An amount of 2.0 M solution of Na2CO3 is boiled in a closed container with excess of CaF2. Very little amount of CaCO3 a...An amount of 0.1millimole of CdSO4 is present in 10 ml acid solution of 0.08 M-HCl. Now H2S is passed to precipitate all...