Ionic EquilibriumHard
Question
Auto-ionisation of liquid NH3 is
2NH3 ⇋NH4+ + NH2-
with KNH3 = [NH4+] [NH2-] = 10-30 at -50oC
Number of amide ions (NH2-), present per mm3 of pure liquid NH3 is
2NH3 ⇋NH4+ + NH2-
with KNH3 = [NH4+] [NH2-] = 10-30 at -50oC
Number of amide ions (NH2-), present per mm3 of pure liquid NH3 is
Options
A.602
B.301
C.200
D.100
Solution
2NH3 ⇋ NH4+ + NH2-
[NH4+] = [NH2-] =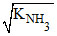 = 10-15 M
= 10-15 M
1 dm3 of solution contains 10-15 moles = 6.02 × 1023 × 10-15 molecules of NH2-.
1 mm3 of solution contains 6.02 × 1023 × 10-15 × 10-6 = 602 NH2- ions.
[NH4+] = [NH2-] =
1 dm3 of solution contains 10-15 moles = 6.02 × 1023 × 10-15 molecules of NH2-.
1 mm3 of solution contains 6.02 × 1023 × 10-15 × 10-6 = 602 NH2- ions.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
There exist an equilibrium between solid BaSO4, Ba2+ and SO42− ions in aqueous medium. Now, if equilibrium is disturbed ...The strength of an aqueous solution of I2 can be determined by titrating the solution with standard solution of:...The acidic hydrolysis of ether (X) shown below is fastest when   Â...If the pH of 0.001 M potassium propionate solution be 8.0, then the dissociation constant of propionic acid will be...Solid Ba (NO3)2 is gradually dissolved in a 1.0 × 10-4 M Na2CO3 solution. At what concentration ofBa2+ will a preci...