Alcohol, Phenol and EtherHard
Question
A solution is found to contain [Cl-]= 1.5 × 10-1M;[Br-] = 5.0 × 10-4M; [CrO42-] = 1.9 × 10-2M. A solution of AgNO3 (100% dissociated) is added to the above solution drop by drop. Which silver salt will precipitate first ?
Given: Ksp(AgCl) = 1.5 × 10-10, Ksp(AgBr) = 5.0 × 10-13, Ksp(Ag2CrO4) = 1.9 × 10-12.
Given: Ksp(AgCl) = 1.5 × 10-10, Ksp(AgBr) = 5.0 × 10-13, Ksp(Ag2CrO4) = 1.9 × 10-12.
Options
A.AgCl
B.AgBr
C.Ag2CrO4
D.AgCl and AgBr together
Solution
For precipitation. [Ag+] ion should be minimum in the solution.
For AgCl: [Ag+]min =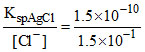 = 10-9 M
= 10-9 M
For AgBr: [Ag+]min =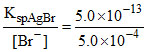
For Ag2CrO4: [Ag+]min
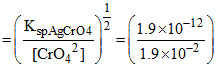 = 10-5 M
= 10-5 M
Therefore, [Ag+] min in solution is in AgCl and AgBr, so both will be precipitated.
For AgCl: [Ag+]min =
For AgBr: [Ag+]min =
For Ag2CrO4: [Ag+]min
Therefore, [Ag+] min in solution is in AgCl and AgBr, so both will be precipitated.
Create a free account to view solution
View Solution FreeMore Alcohol, Phenol and Ether Questions
Match list I with II and then select the correct answer from the codes given below the lists– List I(A) Phenol + NaOH + ...CH3MgBr + E is...Find the correct order :-...The structure of the compounds formed by the reaction of diethyl ether with oxygen of air is -...The compound containing hydrogen bond is-...