Heat and Thermal ExpansionHard
Question
One mole of ideal gas undergoes a cyclic process ACBA as shown in figure. Process AC is adiabatic. The temperatures at A, B and C are 300, 600 and 450 K respectively : - One mole of ideal gas undergoes a cyclic process ACBA as shown in figure. Process AC is adiabatic. The temperatures at A, B and C are 300, 600 and 450 K respectively :
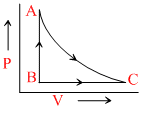

Options
A.In process CA change in internal energy is 225R.
B.In process AB change in internal energy is - 150 R.
C.In process AB change in internal energy is -225 R.
D.Change in internal energy during the whole cyclic process is + 150 R
Solution
Ans (C)
ᐃUCA = nCV(TA - TC)
1 ×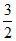 R × (300 - 450) = (- 225R)
R × (300 - 450) = (- 225R)
ᐃUAB = nCV (TB - TA)
1 ×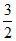 R × (600 - 300) = + 450 R
R × (600 - 300) = + 450 R
ᐃUBC = nCV (TC - TB)
1 ×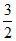 R × (450 - 600) = - 225R
R × (450 - 600) = - 225R
ᐃUABCA = ᐃUCA = ᐃUAB = ᐃUBC = 0
ᐃUCA = nCV(TA - TC)
1 ×
ᐃUAB = nCV (TB - TA)
1 ×
ᐃUBC = nCV (TC - TB)
1 ×
ᐃUABCA = ᐃUCA = ᐃUAB = ᐃUBC = 0
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
The thermo e.m.f. of a thermo-couple is 25 μV/oC at room temperature. A galvanometer of 40 ohm resistance, capable ...In the determination of Young′s modulus by using Searle′s method, a wire of length L = 2mand diameter d = 0....The variation of lengths of two metal rods A and B with change in temperature is shown in figure. The ratio of is : -...Three long straight wires carrying current are arranged mutually parallel as shown in the figure. The force experienced ...The coefficient of apparent expansion of a liquid when determined using two different vessels A and B are γ1 and &#...