Heat and Thermal ExpansionHard
Question
An ideal gas is heated from temperature T1 to T2 under various conditions. The correct statement(s) is are : -
Options
A. ᐃU = nCV (T2 - T1)for isomeric and adiabatic process
B.Work is done at expense of internal energy in an adiabatic process and both have equal values
C.ᐃU = 0for an isothermal process
D.C = 0 for an adiabatic process
Solution
For any process ᐃU + W = nCVᐃT In adiabatic process
Q = ᐃU + W = 0 ⇒ ᐃU = - W
For any process
C =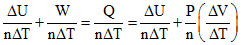
For Q = 0, C = (adiabatic process)
Q = ᐃU + W = 0 ⇒ ᐃU = - W
For any process
C =
For Q = 0, C = (adiabatic process)
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
Ice starts forming in like with water at 0oC when the temperature is - 10oC. If the taken for 1 cm of ice be 7 hours, th...A steel scale measure the length of a copper rod as l0 when both are at 20oC, which is the calibration temperature for t...A sample of gas follows process represented by PV2 = constant. Bulk modulus for this process is B, then which the of the...Three bodies A, B and C have equal surface area and thermal emissivities in the ratio eA : eB : eC = 1 : All the three b...The respective speeds of five molecules are 2, 1, 5, 1, 6, 1.6 and 1.2 km/s. The most probable speed in km/s will be...