Heat and Thermal ExpansionHard
Question
250 g of water and an equal volume of alcohol of mass 200g are placed successively in the same calorimeter and cools from 60oC to 55oC in 130 sec and 67 sec respectively. If the water equivalent of the calorimeter g then the specific heat of alcohol in cal/goC is : -
Options
A.1.30
B.0.67
C.0.62
D.0.985
Solution
Rate of cooling of water = Rate of cooling of alcohol
⇒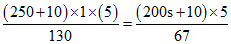
⇒ Specific heat of alcohol
s = 0.62
Rate of cooling of water = Rate of cooling of alcohol
⇒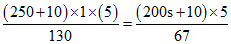
⇒ Specific heat of alcohol
s = 0.62
⇒
⇒ Specific heat of alcohol
s = 0.62
Rate of cooling of water = Rate of cooling of alcohol
⇒
⇒ Specific heat of alcohol
s = 0.62
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
The temperature of a body rises by 44oC when a certain of heat is given t it. The same heat when supplied to 22 g of ice...The following are the P-V diagram for cyclic processes for a gas In which of these processes is heat absorbed by the gas...The coefficient of apparent expansion of a liquid when determined using two different vessels A and B are γ1 and &#...The internal energy of a system remains constant when it undergoes : -...Three identical rods AB, CD and PQ are joined as shown. P and Q are mid points of AB and CD respectively . Ends A, B, C ...