KTGHardBloom L3
Question
During an experiment, an ideal gas is found to obey a condition 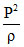 = constant [ρ density of the gas]. The gas is initially at temperature T, pressure P and density ρ . The gas expands such that density changes to
= constant [ρ density of the gas]. The gas is initially at temperature T, pressure P and density ρ . The gas expands such that density changes to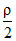
Options
A.The pressure of the gas changes to √2P
B.The temprature of the gas changes to √2P
C.The graph of the above process on the P - T diagram is parabola.
D.The graph of the above process on the P - T diagram is hyperbola.
Solution
PV = nRT
P ∝ ρ T......(i) T ∝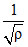 .........(ii) T ∝
.........(ii) T ∝ 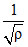 .........(iii)
.........(iii)
P ∝ ρ T......(i) T ∝
Create a free account to view solution
View Solution FreeMore KTG Questions
At constant pressure hydrogen is having temperature of 327o C . Till what temperature it is to be cooled so that the rms...The temperature of ideal gas is the result of which of the following energies :...The mean free path of molecules of a gas, (radius r) is inversely proportional to...The root mean square speed of hydrogen molecules of an ideal hydrogen gas kept in a gas chamber at 0oC is 3180 m/s. The ...Two samples 1 and 2 are initially kept in the same state. The sample 1 is expanded through an isothermal process where a...