Chemical BondingHard
Question
The C - Cl bond length is shortest in :-
Options
A.CH2 = CH - 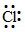
B.CH3 - 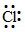
C.C6H5 - 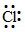
D.C6H5 - CH2 - 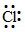
More Chemical Bonding Questions
The molecular shapes of SF4, CF4 and XeF4 are...Which of these is least likely to act as a Lewis base?...NaCl, NaBr, NaI mixture on adding H2SO4 gives gases respectively :-...Which of the following molecular species has unpaired electromn (s) ?...Which of the following molecules/ions does not contain unpaired electrons?...