Surface ChemistryHard
Question
The given graphs / data I, II, III and IV represent general trends observed for different physisorption andchemisorption processes under mild conditions of temperature and pressure. Which of the followingchoice(s) about I, II, III and IV is (are) correct?




Options
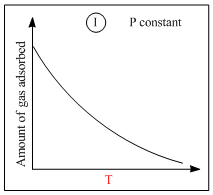
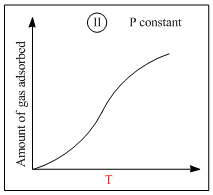
A.I is physisorption and II is chemisorption
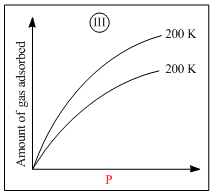
B.I is physisorption and III is chemisorption
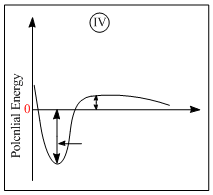
C.IV is chemisorption and II is chemisorption
D.IV is chemisorption and III is chemisorption
Solution
Graph (I) and (III) represent physiosorption because, in physiosorption, the amount of adsorption decreaseswith the increase of temperature and increases with the increase of pressure.
Graph (II) represent chemisorption, because in chemisorption amount of adsorption increase with theincrease of temperature. Graph (IV) is showing the formation of a chemical bond, hence chemisorption
Graph (II) represent chemisorption, because in chemisorption amount of adsorption increase with theincrease of temperature. Graph (IV) is showing the formation of a chemical bond, hence chemisorption
Create a free account to view solution
View Solution FreeMore Surface Chemistry Questions
Platinized asbestos is used as a catalyst in the manufacture of H2SO4. It is an example of...The dispersed phase in colloidal iron(III) hydroxide and colloidal gold is positively and negatively charged, respective...If dispersed phase is liquid and the dispersion medium is solid, the colloid is known as...In Langmuir′s model of adsorption of a gas on a solid surface...A colloid always :...