ThermodynamicsHard
Question
Heat is released by the gas in the process CD. n moles of a monoatomic gas undergoes a cyclic process ABCDA as shown. Processes AB → Isobaric, BC → adiabatic, CD → isochoric, DA → Isothermal. The maximum temperature and minimum temperature in the cycle are 4T0 and T0 respectively. Then
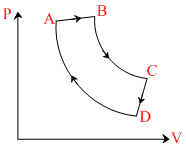

Options
A.TB > TC > TD
B.heat is released by the gas in the process CD.
C.heat is supplied to the gas in the process AB
D.total heat supplied to the gas is zero
More Thermodynamics Questions
An ideal black-body at room temperature is thrown into a furnace. It is observed that...For monoatomic gas the relation between pressure of a gas and temperature T is PαTC where C is. (For adiabatic proc...An increase in temperature (ᐃt) is obtained in adiabatic compression of n moles of an ideal gas. The amount of wor...The work done in the expansion of a gas from a volume 10 cm3 to 20 cm3 in an isobaric process at one atmospheric pressur...Two moles of monoatomic gas are mixed with 1 mole of a diatomic gas. The γγ for the mixture is :...