Heat TransferHard
Question
As part of an experiment to determine the latent heat of vaporisation of water, a student boils some water in a beaker using an electric heater as shown below.
The student notes two sources of error.
Error-1 : thermal energy is lost from the sides of the beaker
Error-2 : as the water is boiling, water splashes out of the beaker.
Which of the following gives the correct effect of these two errors on the calculated value for the specific latent heat ?
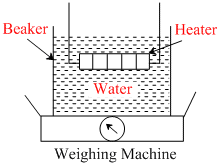
The student notes two sources of error.
Error-1 : thermal energy is lost from the sides of the beaker
Error-2 : as the water is boiling, water splashes out of the beaker.
Which of the following gives the correct effect of these two errors on the calculated value for the specific latent heat ?

Options
A.Error-1 : Increase
Error-2 : Decrease
Error-2 : Decrease
B.Error-1 : Increase
Error-2 : Increase
Error-2 : Increase
C.Error-1 : Decrease
Error-2 : Increase
Error-2 : Increase
D.Error-1 : Decrease
Error-2 : Decrease
Error-2 : Decrease
Solution
(i) Some energy is lost through sides of beakers
(ii) Water splasshes, so mass of water become less
(ii) Water splasshes, so mass of water become less
Create a free account to view solution
View Solution FreeMore Heat Transfer Questions
Two identical square rods of metal are welded end to end as shown in figure (a) 20 calories of heat flows through it in ...The lengths and radii of two rods made of same material are in the ratios 1 : 2 and 2 : 3 respectively. If the temperatu...A long metallic bar is carrying heat from one of its ends to the other end under steady-state. The variation of temperat...A slab of stone of area 0.36 m2 and thickness 0.1 m is exposed on the lower surface to steam at100oC. A block of ice at ...A substance when at high temperature emits wave lenght λ1, λ2, λ3, and λ4 only when this substance i...